Supported Catalysts
Catalysts are essential essential for our society: our bodies and nature would not function without, but also they are key to provide our society with fuels, plastics, drugs, and they are essential to allow the transition from fossil to renewable building blocks. In our group we investigate the most common catalysts: solid (or “heterogenous”) catalyst, which often consist of supported metal nanoparticles.
To optimise the use of often expensive metals, the active phase are typically metal nanoparticles smaller than 10 nm. These active particles are stabilised on porous/high surface area supports, which in turn can be shaped in pellets of extrudates with much larger sizes as generally they have to be thermally mechanically and chemically stable during operation at elevated temperatures and pressures in reactors that are up to several meters or even tens of meters in size.

We we aim to answer fundamental questions related to:
- Fundamentals of catalyst preparation
- Catalyst stability
- Particle size effects
- Impact of metal spatial distribution
- Support effects
- Promoters
For very relevant conversions:
- CO2 to fuels and chemicals
- Ammonia synthesis and decomposition
- Methanol synthesis
- Fischer Tropsch (/clean synthetic fuels)
- Selective oxidation (ethylene and propylene epoxidation, CO PROX, liquid phase oxidation)
- Selective hydrogenation (gas phase and liquid phase)
- Ester hydrogenation

Catalysts allow efficient chemical conversions. As shown in the picture they concentrate reactants, weaken the bonds in molecules by adsorption, stabilise intermediates, and steer the reaction to desorb the desired products. Catalysts are essential to minimise the use of energy and raw materials.
Essential in a catalyst are activity ( for instance measured in turn-over-frequency (how many molecules are converted per second per surface metal atom) or more practically in moles or grams of product produced per gram of catalyst), selectivity (are only the reactants converted that we want to convert? Do we only produce the products that we want to make?) and stability (what is the lifetime of a catalyst?)
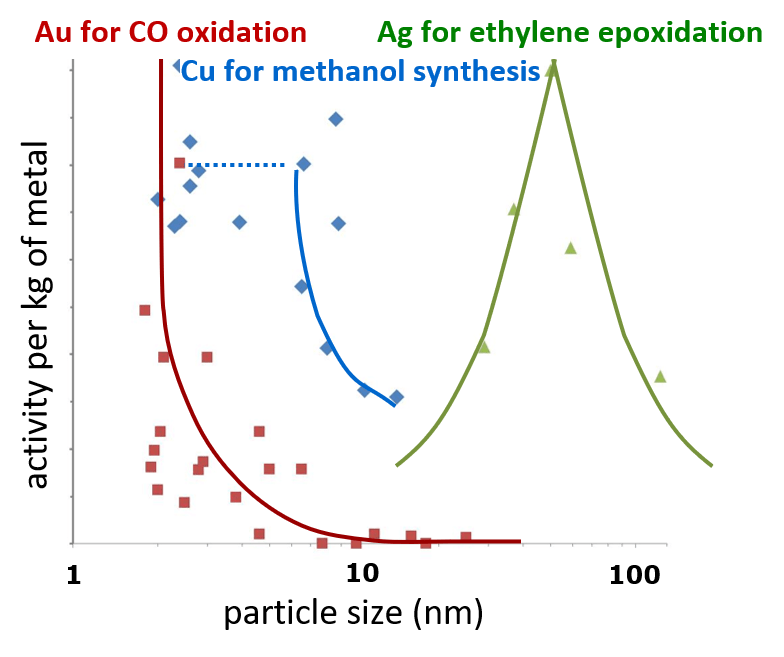
Ag: van Reijen, de Jong(h) et al, J. Catal (2017); Cu: vd Berg et al. Nature Comm. (2016); Au: ; Norskov et al. Nanotoday (2007).